Features:
– Excellent gene delivery efficiency in most cell types including dividing and non-dividing or primary cells.
– Multiple serotypes (AAV1, AAV2, AAV5, AAV6, AAV8, AAV9).
– Integration into chromosome 19 of host genome.
– Induce efficient gene silencing.
Wide Choice of AAV cis Vectors:
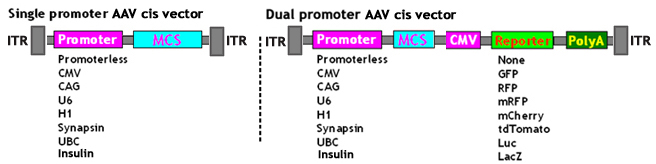
Service Description:
1. Synthesize and clone shRNA into AAV cis vectors.
2. Large scale transfection of AAV·HT™ 293 cells into 2xcell stack.
3. Harvest rAAV followed by purification via advanced 2xCsCl ultra-centrifugation to obtain clinical trial grade of rAAV vector*.
4. Desalting, filter sterilization, and AAV titration via qPCR.
Required Materials: Target gene sequence or validated shRNA sequence.
Turnaround Time: 3 ~ 4 weeks.
Deliverables: >1.0 ml super purified rAAV particle at 1E+13 VG/ml** will be delivered for both in vitro and in vivo applications.
We offer discount for new customer, please request a quote with us today.
We also offer truncated custom shRNA AAV service, please request a quote with us today.
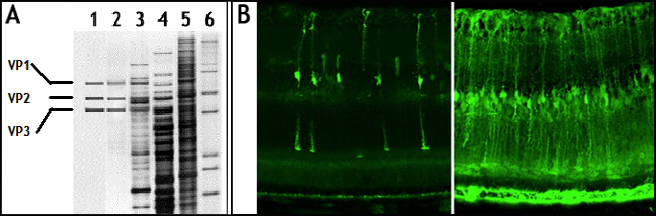
* Figure 1. A comparison of purity and infectivity of rAAV vectors from different sources showing super purified and super infectious (close to clinical trial grade) rAAV vector prepared via our advanced double CsCl ultra-centrifugation approach.
A. rAAV vectors (total 1E+9 VG per lane) from different sources were resolved in SDS-PAGE followed by silver staining. Lane 1: GMP manufactured rAAV vector from CHOP; Lane 2: rAAV prepared via our advanced 2xCsCl ultra-centrifugation approach; Lane 3: rAAV from Vector Core of BCM; Lane 4: rAAV from our competitor “V”; Lane 5: rAAV from our competitor “C”; Lane 6: Protein marker.
B. Super infectious rAAV vector prepared via advanced double CsCl ultra-centrifugation. Left panel: rAAV9-GFP (total 5E+9 VG) from our competitor “V” injected to mouse eye; Right panel: rAAV-9-GFP (total 5E+9 VG) purified via advanced 2xCsCl ultra-centrifugation injected to mouse eye.
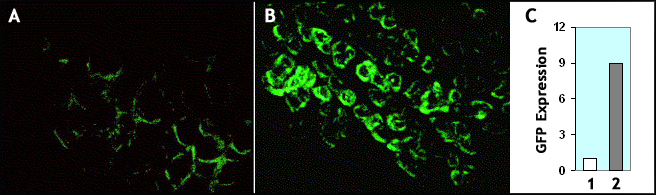
* Figure 2. A comparison of infectivity of rAAV vectors from different sources showing super purified rAAV prepared via advanced double CsCl ultra-centrifugation approach is super infectious. .
rAAV1-GFP (total 2E+9 VG) from different sources were injected to mouse muscle tissue. The GFP fluorescence was visualized 3 weeks post injection. A. rAAV1-GFP from our competitor “V”; B. rAAV1-GFP from our pre-made rAAVs stock purified via advanced double CsCl ultra-centrifugation. C. Quantification data showed that our super purified rAAV (bar 2) is ~ 9 times more infectious than that (bar 1) prepared via conventional CsCl ultra-centrifugation.
** Final viral yield may depend on the nature of transgene. For rAAV serotype 2, we guarantee final deliverable of >2.0 ml at >1E+12 VG/ml.